Patient Information Leaflet: https://www.medicines.org.uk/emc/files/pil.6584.pdf

Stay in the loop with our exclusive offers and new product launches by signing up to our email and SMS updates!




Log in to find out more information about this prescription-only medicine.
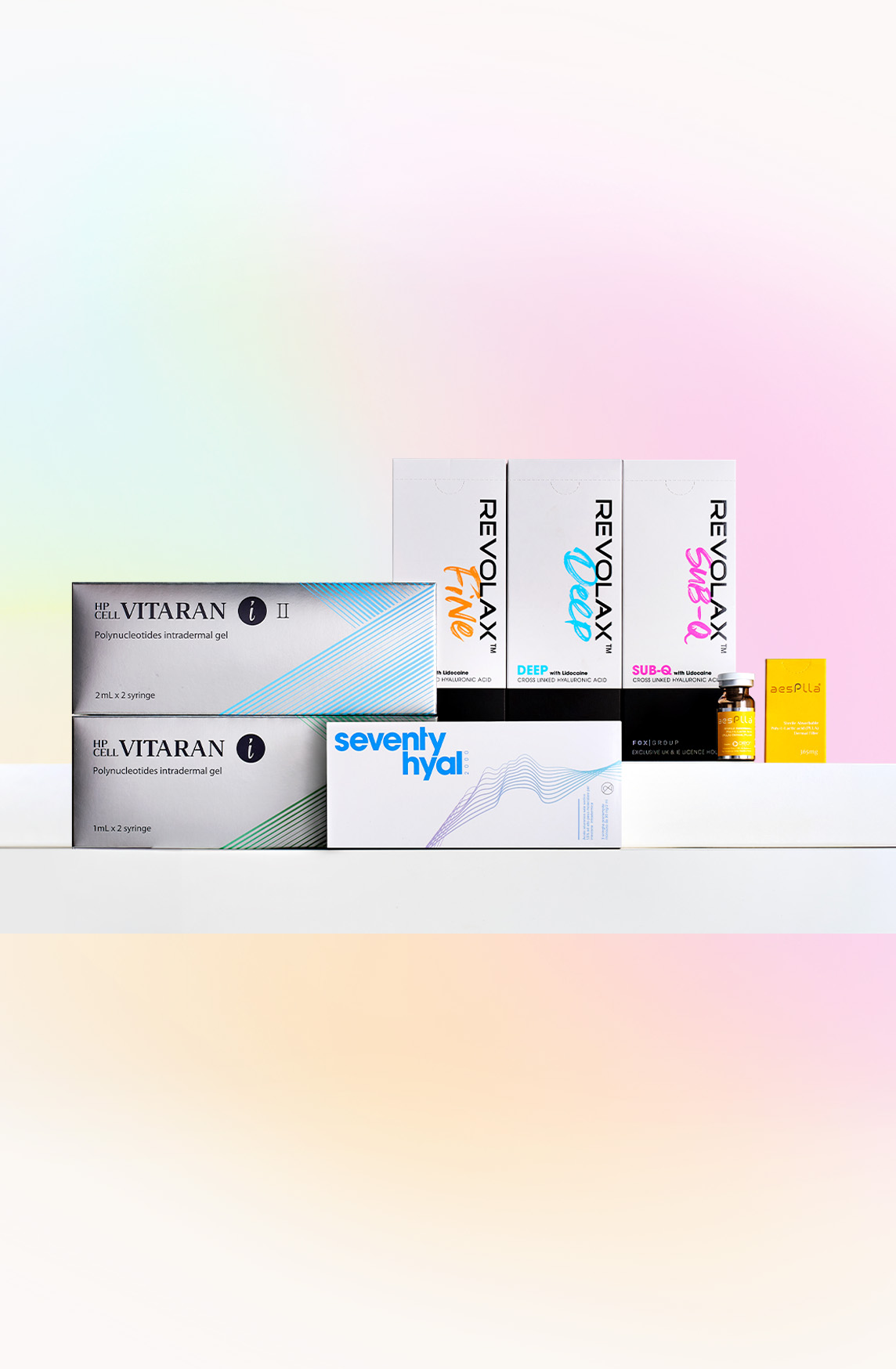
Description
Additional Information
Reviews ( 0 )
EMC: https://www.medicines.org.uk/emc/product/6584
Patient Information Leaflet: https://www.medicines.org.uk/emc/files/pil.6584.pdf
100 g
0 review for Azzalure 125IU (Single Vial)
Join our newsletter for first access to new products, exclusive offers, and more.
Your cart is empty
Looks like you haven't added anything yet.
Prescription ID
Products
Action
#2174 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#26581 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#30058 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#31315 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#31394 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#39517 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#39678 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#39680 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#39681 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#59358 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#77395 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#83874 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#85475 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#89363 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#92963 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#94288 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#102371 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#102372 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#105326 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#105329 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#125611 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#125613 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#127139 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#129243 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#129836 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#132136 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#144393 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#144481 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#144483 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#149542 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#151740 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#164259 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#166408 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#171336 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#176370 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#182706 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#182707 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#191738 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#195277 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#195869 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#195870 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#198482 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#200299 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#201474 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#201668 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#207118 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#218076 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#218558 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#219433 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#223885 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#224312 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#225955 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#227126 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#230252 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#230616 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#234331 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#251016 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#251017 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#261385 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#262776 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#264455 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#267452 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#277214 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#277788 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#279309 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#280002 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#281588 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#290436 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#295740 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311442 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311454 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311455 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311462 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311463 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311464 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311465 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311466 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311467 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311470 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311471 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311472 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311473 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311488 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311490 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311491 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311493 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311494 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311495 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311496 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311497 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311498 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311500 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311501 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311502 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311503 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311504 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311505 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311507 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311508 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311512 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311538 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311540 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311541 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311544 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311545 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311546 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311547 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311548 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311549 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311550 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311551 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311552 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311553 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311554 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311555 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311556 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311557 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311558 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311559 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311560 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311561 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311562 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311563 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311564 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311565 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311566 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311567 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311568 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311569 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311570 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311571 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311572 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311574 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311575 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311576 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311577 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311578 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311579 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311580 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311581 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311582 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311583 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311584 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311585 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311586 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311587 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311588 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311589 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311590 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311591 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311592 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311593 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311594 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311595 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311596 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311597 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311598 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311599 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311600 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311601 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311602 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311603 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311604 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311605 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311606 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311607 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311608 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311609 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311610 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311611 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311612 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311613 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311614 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311615 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311616 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311617 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311618 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311619 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311620 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311625 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311626 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311628 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311675 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311676 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311677 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311680 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311682 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311684 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311686 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311687 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311688 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311689 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311690 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311691 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311692 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311693 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311694 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311695 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311696 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311697 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311698 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311699 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311701 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311702 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311703 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311704 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311705 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311706 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311707 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311708 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311709 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311710 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311711 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311712 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311713 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311714 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311715 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311716 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311717 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311718 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311719 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311720 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311721 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311722 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311723 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311724 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311725 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311726 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311727 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311728 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311729 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311730 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311731 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311732 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311733 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311734 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311735 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311736 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311737 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311738 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311739 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311740 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311741 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311742 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311743 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311744 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311745 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311746 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311747 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311748 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311749 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311750 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311751 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311752 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311753 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311754 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311755 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311756 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311757 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311758 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311759 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311760 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#311761 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#313466 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#320787 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#324260 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#328075 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#330849 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#333869 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#333871 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#334940 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#334941 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#334942 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#335609 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#339224 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#365257 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#378497 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#379055 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#383532 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#385032 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#388405 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#390192 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#396677 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#396950 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#397963 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#405895 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#410764 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#414926 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#415739 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#415743 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#415744 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#418083 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#448453 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#450208 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#454010 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#454011 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#455244 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#457202 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#457846 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#462089 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#463494 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#464216 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#473268 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#473271 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#473272 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#473479 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#474021 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#474744 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#482881 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#503709 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#503712 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#504178 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#506676 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#507793 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#509476 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#513002 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#513007 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#515253 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#522070 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#523142 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#527924 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#528088 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#531271 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#533853 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#534746 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#535223 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#538235 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#541610 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#543265 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#550761 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#552704 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#556617 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#558486 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#558489 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#558776 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#572874 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#576558 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#576728 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|
#589999 -
Products
| Item | Cost | Quantity | Total | |
|---|---|---|---|---|


